Predict whether 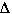 S
is positive, negative, or close to zero for the following changes.
S
is positive, negative, or close to zero for the following changes.
b) 2KClO3(s) ----> 2KCl(s) + 3O2(g)
Answer
In this example 2 moles of reactants form 5 moles of products.
That favors more disorder in the products compared to the reactants. However
the most important change is the production of three moles of gaseous products.
The reactants are solids. Although an equal number of moles of solids are formed,
and the KCl is not as complex as KClO3, the formation of the the
gaseous product produce major disorder. So 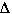 S
is positive for this reaction.
S
is positive for this reaction.