Predict whether 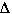 S
is positive, negative, or close to zero for the following changes.
S
is positive, negative, or close to zero for the following changes.
c) 2NO2(g) ----> N2O4(g)
Answer
In this example 2 moles of reactants in the gaseous phase produce
1 mole of product in the gaseous phase. even though the products are more complex
the system is becoming more ordered and 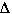 S
is negative.
S
is negative.