Thank you for completing this ACA. You should print this page out and bring it to class so if you have any questions you can ask them.
ACA Response Page
It is OK to use your textbook, but if you can
answers the questions without it that is OK too.
I recommend you print out this page and bring it to class.
, here are your responses
to the ACA and the Expert's response.
1. Draw the Lewis structure for each of the following compounds,
then complete the table; (NOTE: You may wish to consult your textbook to determine the molecular geometry and the terminal atom-central atom-terminal atom bond angle. Additionally you may want to look at the Models360 website we used in class.)
Formula |
# of valence electrons (molecule/ion) |
# of bonding domains on CA(s) |
# of nonbonding domains on CA(s) |
Molecular Geometry |
TA-CA-TA bond angle |
SO2
|
(18)
|
(2)
|
(1)
|
(bent) |
(>120 degrees)
|
SO32–
|
(26)
|
(3)
|
|
|
(>109.5 degrees)
|
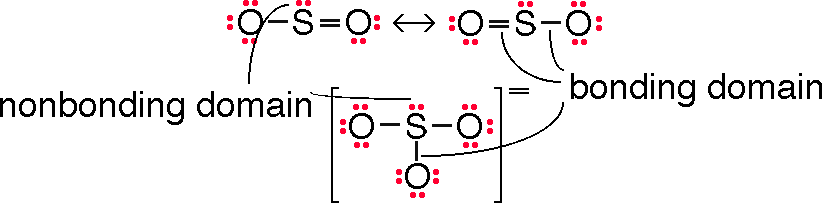
2a. What is the bond order (how many bonds) of the S-O bond
in SO2?(Note: a single bond has a bond order of 1, a double bond has a bond order
of 2, and a triple bond has a bond order of 3.)
(The bond order for S-O bond is 1.5. There are two resonance structures to represent the Lewis structure for SO2. So the S–O bond order is 1.5.)
b. How many Lewis structures are needed to represent the
bonding in SO2?
(Two structures are needed. See figure in Q1.)
3a. What is the formal charge (page 383) on the sulfur atom in SO2 in your structure?
(In the resonance structures draw for SO2 in Q1 the formal charge on S is
FC(S) = 6 – 0.5*6 – 2 = +1)
b. If the formal charge on the sulfur atom is not 0 in your answer to Question 3a, how can you reduce the formal charge on the sulfur atom to get it to zero?
(Using the Lewis structure below
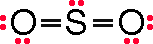
the formal charge on sulfur is reduced to 0. FC(S) = 6 - 0.5*8 – 2 = 0. Since sulfur is in the third period it can have more than eight electrons on the sulfur in this Lewis structure. So by moving a nonbonding pair from an oxygen atom the formal charge is reduced to 0.)
Answer the following questions using bond energies or Table 9.2 on page 353 in your textbook:
4. How much energy (kJ mol-1) is required to break,
CO bond in CO2? |
(803 kJ mol-1) |
FF bond in F2? |
(155 kJ mol-1) |
CF bond in CF4? |
(485 kJ mol-1) |
OF bond in OF2? |
|
5. Calculate  H˚ (kJ mol-1) for the reaction,
H˚ (kJ mol-1) for the reaction,
CO2(g) + 3F2(g) -----> CF4(g) + OF2(g)
( H˚ = BE(reactants) – BE(Products)
H˚ = BE(reactants) – BE(Products)
= 2 BE(C=O) + 3 BE(F–F) –[4 BE(C–F) + 2 BE(O–F)
= 2 * (803 kJ mol-1) + 3 * (155 kJ mol-1) – [4 * (485 kJ mol-1) + 2 * (190 kJ mol-1)
= –249 kJ mol-1)
6. Write the formation equation for PCl3(g).
(1/4P4(s) + 3/2Cl2(g)
----> PCl3(g))
7. Is there anything about the questions that you feel you do not understand?
List your concerns/questions.
8. If there is one question you would like to have answered in lecture, what
would that question be?
