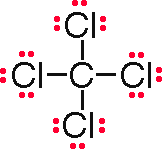
Formula |
Symbol of Central Atom(s) (CA) |
Symbol of Terminal Atom(s) |
# of valence electrons on a C atom |
# of valence electrons on a Cl atom |
total # of valence electrons on CCl4 |
CCl4 |
|
|
|
|
|

For C in CCl4: C has 4 valence electrons, 8 bonding electrons and 0 nonbonding electrons. FC (C) = 4 – 0.5*8 – 0 = 0 For Cl in CCl4: Cl has 7 valence electrons, 2 bonding electrons and 6 nonbonding electrons. FC (Cl) = 7 – 0.5*2 – 6 = 0 |
|---|
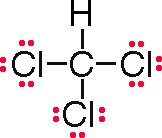
Formula |
Formal Charge on C |
Formal Charge on H |
Formal Charge on Cl |
CHCl3 |
|
|
|
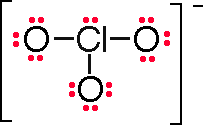
Formula |
Formal Charge on Cl |
Formal Charge on O |
ClO3– |
|
|
H-H bond in H2?
(436)Cl-Cl bond in Cl2?
(242)H-Cl bond in HCl?
(431)
H-H bond in H2?
(-436)Cl-Cl bond in Cl2?
(-242)H-Cl bond in HCl?
(-431)